|
|
| (21 intermediate revisions by 3 users not shown) |
| Line 1: |
Line 1: |
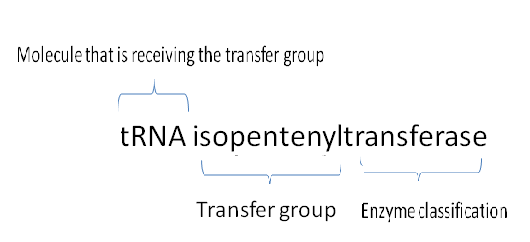
| Molecular function and biological processes of this enzyme was obtained from the ProKnow server.
| | '''Function from its name''' |
| Molecular function of this enzyme is ATP binding. This means that the enzyme interacts selectively with ATP at the 5'end and acts as a universally important coenzyme and enzyme regulator.
| |
| On the other hand, the biological process of this enzyme is tRNA modification. This involves the covalent alteration of one or more nucleotides within the tRNA, thus leading to a change in the properties of the tRNA.
| |
|
| |
|
| '''Molecular Function'''
| | [[Image:reaction1.png|framed|center|none]] |
|
| |
|
| | | [[How the enzyme act on tRNA| Next]] |
| [[Image:PROKNOW2- molecular function.png]] | |
| | |
| | |
| '''Biological Process'''
| |
| | |
| | |
| [[Image:PROKNOW-biological process.png]]
| |
| | |
| | |
| ==Localisation of Expression==
| |
| [[Image:Mouse gene atlas.png]]
| |
| | |
| | |
| [[Image:Human geneAtlas.png]]
| |
| | |
| | |
| using the motif search, The C2-H2 Zinc finger motif found in tRNA-IPT also contributes to the enzyme's function. This motif is commonly found in nucleic acid-binding proteins and is composed of 25 to 30 amino acid residues.
| |
| | |
| Motif ZINC_FINGER_C2H2_1 in your sequence
| |
| | |
| Prosite ID:
| |
| ZINC_FINGER_C2H2_1 (PS00028)
| |
| | |
| Description:
| |
| Zinc finger C2H2 type domain signature.
| |
| | |
| Pattern:
| |
| C-x(2,4)-C-x(3)-[LIVMFYWC]-x(8)-H-x(3,5)-H.
| |
| | |
| Appearance:
| |
| Position Found Motif
| |
| 396..418 CDLCDRIIIGDREWAAHIKSKSH
| |
| | |
| Sequence:
| |
| MASVAAARAVPVGSGLRGLQRTLPLVVILGATGTGKSTLALQLGQRLGGEIVSADSMQVE
| |
| GLDIITNKVSAQEQRICRHHMISFVDPLVTNYTVVDFRNRATALIEDIFARDKIPIVVGG
| |
| TNYYIESLLWKVLVNTKPQEMGTEKVIDRKVELEKEDGLVLHKRLSQVDPEMAAKLHPHD
| |
| KRKVARSLQVFEETGISHSEFLHRQHTEEGGGPLGGPLKFSNPCILWLHADQAVLDERLD
| |
| KRVDDMLAAGLLEELRDFHRRYNQKNVSENSQDYQHGIFQSIGFKEFHEYLITEGKCTLE
| |
| TSNQLLKKGIEALKQVTKRYARKQNRWVKNRFLSRPGPIVPPVYGLEVSDVSKWEESVLE
| |
| PALEIVQSFIQGHKPTATPIKMPYNEAENKRSYHLCDLCDRIIIGDREWAAHIKSKSHLN
| |
| QLKKRRRLDSDAVNTIESQSVSPDHNKEPKEKGSPGQNDQELKCSV
| |
| | |
| | |
| | |
| | |
| | |
| A schematic representation of a zinc finger domain is shown below:
| |
| x x
| |
| x x
| |
| x x
| |
| x x
| |
| x x
| |
| x x
| |
| C H
| |
| x \ / x
| |
| x Zn x
| |
| x / \ x
| |
| C H
| |
| x x x x x x x x x x
| |
| | |
| | |
| http://ca.expasy.org/cgi-bin/nicedoc.pl?PDOC00028
| |
| | |
| GENOMIC CONTEXT
| |
| In bacteria, the gene encoding this enzyme is known as miaA while the human and S. cerevisaea is TRIT1 and mod5 respectively. Although gene name differ in different organisms, they all synthesize enzyme of the same function.
| |
| | |
| ==Sequence Motif==
| |
| | |
| [[Image:motif.jpg]]
| |
| | |
| ==Functional partners of tRNA isopentenyl transferase==
| |
| | |
| [[Image:protein interaction.jpg]]
| |
| | |
| * Input protein - ''miaA'' : tRNA delta(2)-isopentenylpyrophosphate transferase (IPP transferase) (Isopentenyl-diphosphate-tRNA isopentenyltransferase) (IPTase) (IPPT) (314 aa)(Bacillus halodurans)
| |
| | |
| | |
| * ''mutL'' : DNA mismatch repair protein mutL (637 aa)
| |
| | |
| * ''hfq'' : Protein hfq {UniProtKB/Swiss-Prot-Q9KAC4} (78 aa)
| |
| | |
| * ''mutS'' : DNA mismatch repair protein mutS (865 aa)
| |
| | |
| * ''BH2367'' : BH2367 protein {UniProtKB/TrEMBL-Q9KAC2} (256 aa)
| |
| | |
| * ''BH2362'':BH2362 protein {UniProtKB/TrEMBL-Q9KAC7} (418 aa)
| |
| | |
| * ''BH3154'' : Protease specific for phage lambda cII repressor (310 aa)
| |
| | |
| * ''BH3155'': Protease specific for phage lambda cII repressor (319 aa)
| |
| | |
| * ''BH0545'' :BH0545 protein {UniProtKB/TrEMBL-Q9KFD6} (157 aa)
| |
|
| |
| * ''BH1020'' : BH1020 protein {UniProtKB/TrEMBL-Q9KE38} (391 aa)
| |
| | |
| * dapF : Diaminopimelate epimerase (EC 5.1.1.7) (DAP epimerase) (286 aa)
| |