Result of SNAPG: Difference between revisions
No edit summary |
|||
| Line 1: | Line 1: | ||
== SNAPG Structure == | == SNAPG Structure == | ||
=== Structure architecture === | === Structure architecture === | ||
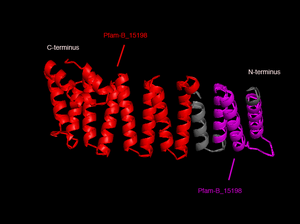
SNAPG protein structure obtained from ''Danio rerio'' organism comprises of 4 molecular chains (Chain A, B, C and D) interacting to one another via non-bonded contacts. Each chain secondary structure | SNAPG protein structure obtained from ''Danio rerio'' organism comprises of 4 identical molecular chains (Chain A, B, C and D) interacting to one another via non-bonded contacts. Each chain is 307 amino acid residues in length and made of 69% helical secondary structure of 16 helices, 34 helix-helix interaction, 8 beta turns and 2 gamma turns [http://www.ebi.ac.uk/thornton-srv/databases/cgi-bin/pdbsum/GetPage.pl?pdbcode=2ifu&template=protein.html&o=SUMMARY&l=1&c=1&chain=A]. | ||
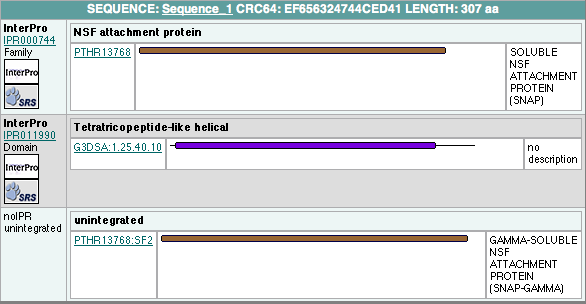
In order to analyze protein structure of SNAPG, structural comparison to known protein structure is required. An insight to SNAPG structural arrangement provides various informative data on possible protein functions and interactions with another protein and/or DNA. Based on protein families database, Pfam at Sanger, it was found that SNAPG protein matched to Pfam-B protein families and consist of 2 domains, Pfam-B_7270 (PB007270) and Pfam-B_15198 (PB015189) respectively as shown in Figure 1. Both of 2 domains appears to be associated with NSF attachment protein activity [http://www.ebi.ac.uk/interpro/IEntry?ac=IPR000744]. | In order to analyze protein structure of SNAPG, structural comparison to known protein structure is required. An insight to SNAPG structural arrangement provides various informative data on possible protein functions and interactions with another protein and/or DNA. Based on protein families database, Pfam at Sanger, it was found that SNAPG protein matched to Pfam-B protein families and consist of 2 domains, Pfam-B_7270 (PB007270) and Pfam-B_15198 (PB015189) respectively as shown in Figure 1. Both of 2 domains appears to be associated with NSF attachment protein activity [http://www.ebi.ac.uk/interpro/IEntry?ac=IPR000744]. | ||
| Line 9: | Line 9: | ||
[[Image:2ifuA interpro.png]] | [[Image:2ifuA interpro.png]] | ||
=== Structural comparison === | === Structural comparison === | ||
Revision as of 22:32, 10 June 2007
SNAPG Structure
Structure architecture
SNAPG protein structure obtained from Danio rerio organism comprises of 4 identical molecular chains (Chain A, B, C and D) interacting to one another via non-bonded contacts. Each chain is 307 amino acid residues in length and made of 69% helical secondary structure of 16 helices, 34 helix-helix interaction, 8 beta turns and 2 gamma turns [1].
In order to analyze protein structure of SNAPG, structural comparison to known protein structure is required. An insight to SNAPG structural arrangement provides various informative data on possible protein functions and interactions with another protein and/or DNA. Based on protein families database, Pfam at Sanger, it was found that SNAPG protein matched to Pfam-B protein families and consist of 2 domains, Pfam-B_7270 (PB007270) and Pfam-B_15198 (PB015189) respectively as shown in Figure 1. Both of 2 domains appears to be associated with NSF attachment protein activity [2].
Structural comparison
Dali webserver is one of the powerful tool to screen any protein that are structurally homologous with our query. Two structurally related proteins with highest Z-value generated by Dali server were chosen for SNAPG structure comparison analysis. These proteins were vesicular transport ptotein sec17 (1qqe) and type 4 fimbrial biogenesis protein (2f17) (refer to table..)
| PDB-chain | Structure | Z-value | % identity | Protein |
|---|---|---|---|---|
| 2IFU-A | 37.8 | 100 | Endocytosis/exocytosis. Gamma-SNAP (Danio rerio) | |
| 1QQE-A | 23.3 | 23 | Protein binding. Vesicular transport protein sec17(yeast) | |
| 2FI7-A | 12.9 | 14 | Protein transport. Type 4 fimbrial biogenesis protein pili (Pseudomonas aeruginosa) |