Arylformamidase Structure: Difference between revisions
No edit summary |
No edit summary |
||
| Line 138: | Line 138: | ||
''Blue indicates semi-conservation'' | ''Blue indicates semi-conservation'' | ||
[[Image:Untitled2.PNG]] | |||
This image shows the triad conserved residues in our protein | |||
SER 136 | |||
HIS 241 | |||
GLU 214 | |||
The unknown ligand is shown protruding from a groove and kinda neatly fits in the pocket. | |||
[[Arylformamidase | Return to the main page...]] | [[Arylformamidase | Return to the main page...]] | ||
Revision as of 05:48, 28 May 2008
1. Structure of Arylformamidase
Arylformamidase
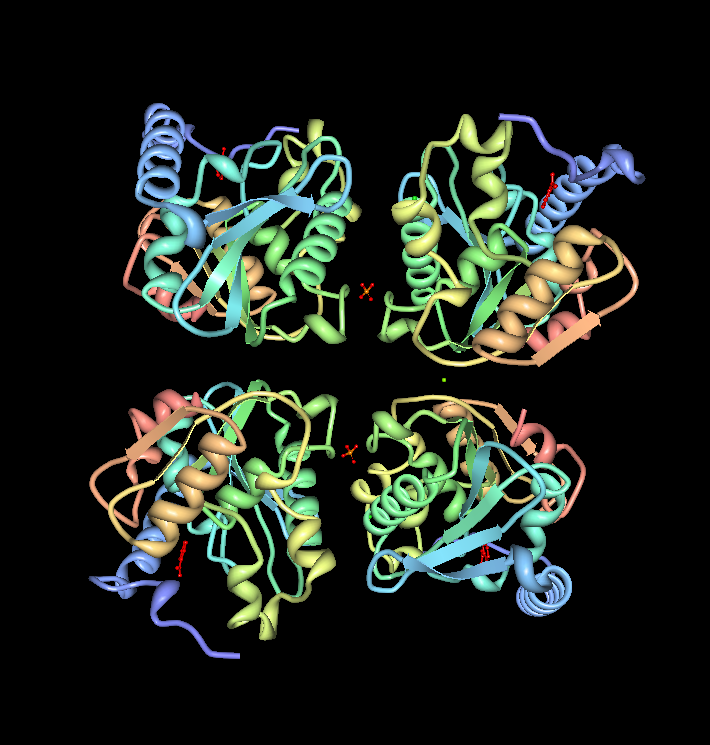
The image above shows the chains A (upper right), B (upper left), C (lower right) & D (lower left) interacting. The molecules in the middle of chains A & B and chains C & D is phosphate ion (PO4). The green molecule between chain B & D is a magnesium ion (Mg). These ions aren't biologically significant and could only be an artefact. Those chains exist as indivitual functional units.
Chain A of arylformamidase
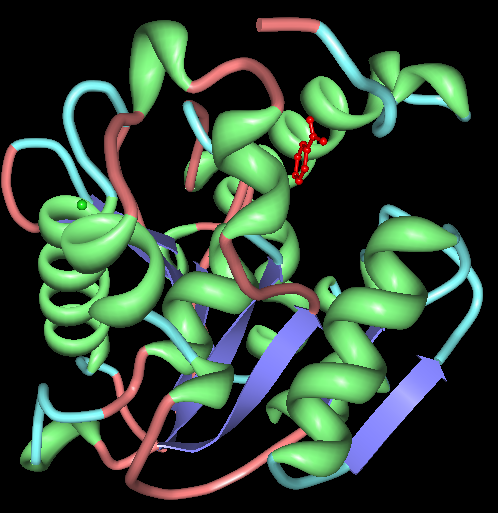
The red molecule in the middle is an unknown ligand containing a ring composed of 9 oxygen molecules. The green sphere is a chloride ion.
2. Putative Thioesterase
Thioesterases split ester groups into acid and alcohol in the presence of water, specifically at a thiol group.
Thioesterases or thiolester hydrolases are members of E.C.3.1.2.
Our structure resembles carboxylesterases (hydrolases) hence it is predicted that it has similar cellular activity.
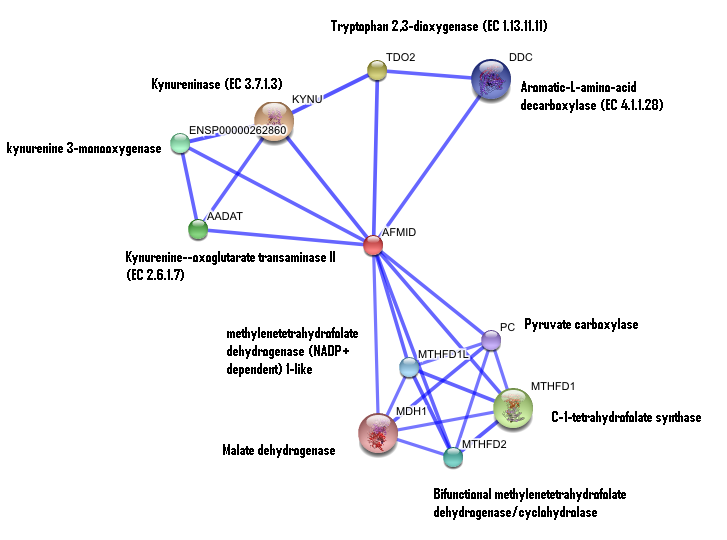
3. Interaction of human arylformamidase (AFMID) with other proteins
The interaction between the proteins have been determined from curated STRING database (significant score). However there is no significant evidence for:
1- Neighborhood in the genome
2- Gene fusions
3- Cooccurence across genomes
4- Co-Expression
5- Experimental/Biochemical data
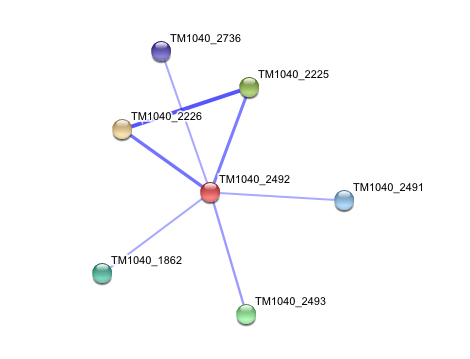
4. Interaction of Silicibacter Sp. arylformamidase (AFMID) with other proteins
TM1040_2226 Tryptophan 2,3-dioxygenase (279 aa)
TM1040_2225 Kynureninase (396 aa)
TM1040_2493 Succinic semialdehyde dehydrogenase (490 aa)
TM1040_1862 Hypothetical protein (212 aa)
TM1040_2491 Creatinase (402 aa)
TM1040_2736 Transketolase, putative (794 aa)
There is no significant evidence for these interactions (score= ~0.5)
4. DALI OUTPUT
Metagenomic Archea Carboxylesterase A chain
PDB link title
Archaeoglobus fulgidus Carboxylesterase (Chains A B C D)
File:Carboxylesterase (archaeon).txt
PDB link title
These chains exist as monomers (from literature). Hence it is expected that our protein exists as a monomer but during crystalization it interacts with its chains.
5. Secondary structure analysis
PDBSum output for arylformamidase
PDBSUM [1]
Archeon Carboxylesterase secondary structure
6. The conservation of the ser/his/asp catalytic triad
Yellow indicates conservation
Blue indicates semi-conservation
This image shows the triad conserved residues in our protein
SER 136
HIS 241
GLU 214
The unknown ligand is shown protruding from a groove and kinda neatly fits in the pocket.