Pyridoxal Phosphatase Introduction: Difference between revisions
No edit summary |
No edit summary |
||
| Line 3: | Line 3: | ||
The biological significance of protein phosphatases - their structures, rather - are the information provided pertaining to both normal and pathophysiologic processes, including transcription regulation, regulation of major signalling pathways and type 1 diabetes. These studies could contribute significantly towards human disease studies, the development of treatment methods for microbial infections, and ultimately bringing cellular and molecular biology studies to a whole new level.<BR> | The biological significance of protein phosphatases - their structures, rather - are the information provided pertaining to both normal and pathophysiologic processes, including transcription regulation, regulation of major signalling pathways and type 1 diabetes. These studies could contribute significantly towards human disease studies, the development of treatment methods for microbial infections, and ultimately bringing cellular and molecular biology studies to a whole new level.<BR> | ||
Protein phosphatases, in general, have a distinctive catalytic domain comprised of a beta sheet | Protein phosphatases, in general, have a distinctive catalytic domain comprised of a beta sheet sandwiched between alpha helices. The active site contains a magnesium ion that assists in the phosphate-cleaving reaction. In the case of Pyridoxal Phosphatase, a large domain covers the the active site, and this is typical for phosphatases that act on small molecules. Extra domains are therefore present to create a tighter active site. <BR> | ||
'''Chronophin''' <BR> | '''Chronophin''' <BR> | ||
Revision as of 14:00, 31 May 2008
An Introduction to Protein Phosphatases
The biological significance of protein phosphatases - their structures, rather - are the information provided pertaining to both normal and pathophysiologic processes, including transcription regulation, regulation of major signalling pathways and type 1 diabetes. These studies could contribute significantly towards human disease studies, the development of treatment methods for microbial infections, and ultimately bringing cellular and molecular biology studies to a whole new level.
Protein phosphatases, in general, have a distinctive catalytic domain comprised of a beta sheet sandwiched between alpha helices. The active site contains a magnesium ion that assists in the phosphate-cleaving reaction. In the case of Pyridoxal Phosphatase, a large domain covers the the active site, and this is typical for phosphatases that act on small molecules. Extra domains are therefore present to create a tighter active site.
Chronophin
Chronophin is a phosphatase (belonging to the HAD family) that is involved in two different biological actions:
- Regulation of the actin cytoskeleton
- Vitamin B6 metabolism
In the context of Vitamin B6 metabolism, Chronophin undertakes this function under the name of Pyridoxal Phosphatase. Chronophin's main role in Vitamin B6 metabolism is the phosphorylation of pyridoxal-5'-phosphate(PLP, or the coenzymatically active form of vitamin B6), and this is part of a complicated pathway for degradation of the vitamin.
Almo et al., 2007 mentions that the PDB code for Chronophin is 2oyc. This PDB code will be appearing rather regularly in the structural component of this paper. Of similar importance is the PLP-bound form of Chronophin, which is obtained by substituting the Magnesium ions for catalytically inert Calcium ions. This affects the catalyic aspartic acid (Asp25), resulting in compete inactivation of the enzyme. An example of the PLP-bound form of Chronophin is also known as Pyridoxal-5'-Phosphate Phosphatase (PDB ID: 2cft), which like 2oyc, will be mentioned further down the paper.
2cfsA
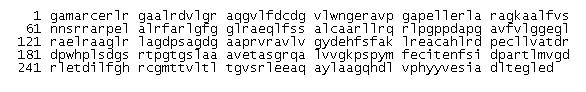
Sequence for the protein of interest, Pyridoxal Phosphatase
Sequence for the human homolog of Pyridoxal Phosphatase